Abstract
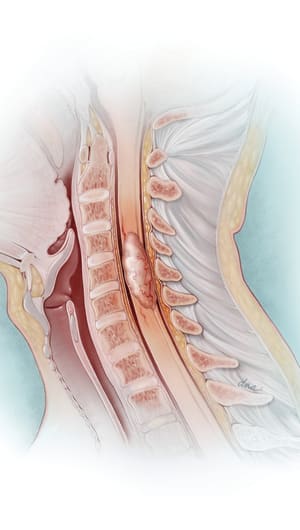
Myelofibrosis is a myeloproliferative neoplasm characterized by the buildup of fibrous scar tissue in the bone marrow occurring secondary to the secretion of inflammatory cytokines, leading to cytopenias, dysfunctional hematopoiesis, and constitutional symptoms. One of the pathologic mechanisms that underlies myelofibrosis is aberrant activation of the Janus kinase (JAK)-STAT pathway. Targeting the JAK-STAT pathway via JAK inhibition can lead to significant improvements in spleen volume reduction and symptom improvement in intermediate- and high-risk myelofibrosis. The first JAK inhibitor approved by the US Food & Drug Administration was ruxolitinib in 2011. Recently, there have been additional JAK inhibitors approved for myelofibrosis, including fedratinib, pacritinib, and momelotinib. The emergence of these new therapies offers additional treatment options for patients with myelofibrosis. This article reviews the pharmacology, efficacy, safety, dosing, administration, and implications for advanced practitioners of newer JAK inhibitors (fedratinib, pacritinib, and momelotinib) in the treatment of myelofibrosis.
References
Baek, D. W., Cho, H. J., Lee, J. M., Kim, J., Moon, J. H., & Sohn, S. K. (2022). Light and shade of ruxolitinib: Positive role of early treatment with ruxolitinib and ruxolitinib withdrawal syndrome in patients with myelofibrosis. Expert Review of Hematology, 15(7), 573–581. https://doi.org/10.1080/17474086.2022.2088499
Bose, P., & Verstovsek, S. (2020). Management of myelofibrosis after ruxolitinib failure. Leukemia and Lymphoma, 61(8), 1797–1809. https://doi.org/10.1080/10428194.2020.1749606
Gangat, N., & Tefferi, A. (2020). Myelofibrosis biology and contemporary management. British Journal of Haematology, 191(2), 152–170. https://doi.org/10.1111/bjh.16576
Gerds, A. T., Savona, M. R., Scott, B. L., Talpaz, M., Egyed, M., Harrison, C. N.,…Mascarenhas, J. O. (2020). Determining the recommended dose of pacritinib: Results from the PAC203 dose-finding trial in advanced myelofibrosis. Blood Advances, 4(22), 5825–5835. https://doi.org/10.1182/bloodadvances.2020003314
Gerds, A. T., Verstovsek, S., Vannucchi, A. M., Al-Ali, H. K., Lavie, D., Kuykendall, A. T.,…Mesa, R. (2023). Momelotinib versus danazol in symptomatic patients with anaemia and myelofibrosis previously treated with a JAK inhibitor (MOMENTUM): An updated analysis of an international, double-blind, randomised phase 3 study. Lancet Haematology, 10(9), e735–e746. https://doi.org/10.1016/S2353-3026(23)00174-6
Guglielmelli, P., Ghirardi, A., Carobbio, A., Masciulli, A., Maccari, C., Mora, B.,…Barbui, T. (2022). Impact of ruxolitinib on survival of patients with myelofibrosis in the real world: update of the ERNEST Study. Blood Advances, 6(2), 373–375. https://doi.org/10.1182/bloodadvances.2021006006
Harrison, C. N., Schaap, N., Vannucchi, A. M., Kiladjian, J. J., Jourdan, E., Silver, R. T.,…Mesa, R. A. (2020). Fedratinib in patients with myelofibrosis previously treated with ruxolitinib: An updated analysis of the JAKARTA2 study using stringent criteria for ruxolitinib failure. American Journal of Hematology, 95(6), 594–603. https://doi.org/10.1002/ajh.25777
Harrison, C. N., Schaap, N., Vannucchi, A. M., Kiladjian, J. J., Tiu, R. V., Zachee, P.,…Mesa, R. A. (2017). Janus kinase-2 inhibitor fedratinib in patients with myelofibrosis previously treated with ruxolitinib (JAKARTA-2): A single-arm, open-label, non-randomised, phase 2, multicentre study. Lancet Haematology, 4(7), e317–e324. https://doi.org/10.1016/S2352-3026(17)30088-1
Harrison, C. N., Vannucchi, A. M., Platzbecker, U., Cervantes, F., Gupta, V., Lavie, D.,…Verstovsek, S. (2018). Momelotinib versus best available therapy in patients with myelofibrosis previously treated with ruxolitinib (SIMPLIFY 2): A randomised, open-label, phase 3 trial. Lancet Haematology, 5(2), e73–e81. https://doi.org/10.1016/S2352-3026(17)30237-5
Jain, T., & Mesa, R. (2016). The development, safety and efficacy of pacritinib for the treatment of myelofibrosis. Expert Review of Anticancer Therapy, 16(11), 1101–1108. https://doi.org/10.1080/14737140.2016.1233061
Maffioli, M., Mora, B., Ball, S., Iurlo, A., Elli, E. M., Finazzi, M. C.,…Passamonti, F. (2022). A prognostic model to predict survival after 6 months of ruxolitinib in patients with myelofibrosis. Blood Advances, 6(6), 1855–1864. https://doi.org/10.1182/bloodadvances.2021006889
Mascarenhas, J. (2022). Pacritinib for the treatment of patients with myelofibrosis and thrombocytopenia. Expert Reviews in Hematology, 15(8), 671–684. https://doi.org/10.1080/17474086.2022.2112565
Mascarenhas, J., Hoffman, R., Talpaz, M., Gerds, A. T., Stein, B., Gupta, V.,…Verstovsek, S. (2018). Pacritinib vs best available therapy, including ruxolitinib, in patients with myelofibrosis: A randomized clinical trial. JAMA Oncology, 4(5), 652–659. https://doi.org/10.1001/jamaoncol.2017.5818
Mesa, R. A., Gotlib, J., Gupta, V., Catalano, J. V., Deininger, M. W., Shields, A. L.,…Verstovsek, S. (2013). Effect of ruxolitinib therapy on myelofibrosis-related symptoms and other patient-reported outcomes in COMFORT-I: A randomized, double-blind, placebo-controlled trial. Journal of Clinical Oncology, 31(10), 1285–1292. https://doi.org/10.1200/JCO.2012.44.4489
Mesa, R. A., Kiladjian, J. J., Catalano, J. V., Devos, T., Egyed, M., Hellmann, A.,…Gotlib, J. (2017b). SIMPLIFY-1: A phase III randomized trial of momelotinib versus ruxolitinib in janus kinase inhibitor-naive patients with myelofibrosis. Journal of Clinical Oncology, 35(34), 3844–3850. https://doi.org/10.1200/JCO.2017.73.4418
Mesa, R. A., Vannucchi, A. M., Mead, A., Egyed, M., Szoke, A., Suvorov, A.,…Harrison, C. N. (2017a). Pacritinib versus best available therapy for the treatment of myelofibrosis irrespective of baseline cytopenias (PERSIST-1): An international, randomised, phase 3 trial. Lancet Haematology, 4(5), e225–e236. https://doi.org/10.1016/S2352-3026(17)30027-3
Mullally, A., Hood, J., Harrison, C., & Mesa, R. (2020). Fedratinib in myelofibrosis. Blood Advances, 4(8), 1792–1800. https://doi.org/10.1182/bloodadvances.2019000954
National Comprehensive Cancer Network. (2023). NCCN Clinical Practice Guidelines in Oncology: Myeloproliferative Neoplasms. V1.2023. https://www.nccn.org/professionals/physician_gls/pdf/mpn.pdf
Newberry, K. J., Patel, K., Masarova, L., Luthra, R., Manshouri, T., Jabbour, E.,…Verstovsek, S. (2017). Clonal evolution and outcomes in myelofibrosis after ruxolitinib discontinuation. Blood, 130(9), 1125–1131. https://doi.org/10.1182/blood-2017-05-783225
Oh, S. T., Mesa, R. A., Harrison, C. N., Bose, P., Gerds, A. T., Gupta, V.,…Verstovsek, S. (2023). Pacritinib is a potent ACVR1 inhibitor with significant anemia benefit in patients with myelofibrosis. Blood Advances, 7(19), 5835–5842. https://doi.org/10.1182/bloodadvances.2023010151
Palandri, F., Breccia, M., Bonifacio, M., Polverelli, N., Elli, E. M., Benevolo, G.,…Vianelli, N. (2020). Life after ruxolitinib: Reasons for discontinuation, impact of disease phase, and outcomes in 218 patients with myelofibrosis. Cancer, 126(6), 1243–1252. https://doi.org/10.1002/cncr.32664
Palandri, F., Palumbo, G. A., Elli, E. M., Polverelli, N., Benevolo, G., Martino, B.,…Bonifacio, M. (2021). Ruxolitinib discontinuation syndrome: incidence, risk factors, and management in 251 patients with myelofibrosis. Blood Cancer Journal, 11(1), 4. https://doi.org/10.1038/s41408-020-00392-1
Pardanani, A., Harrison, C., Cortes, J. E., Cervantes, F., Mesa, R. A., Milligan, D.,…Tefferi, A. (2015). Safety and efficacy of fedratinib in patients with primary or secondary myelofibrosis: a randomized clinical trial. JAMA Oncology, 1(5), 643–651. https://doi.org/10.1001/jamaoncol.2015.1590
Passamonti, F., & Mora, B. (2023). Myelofibrosis. Blood, 141(16), 1954–1970. https://doi.org/10.1182/blood.2022017423
Reynolds, S. B., & Pettit, K. (2022). New approaches to tackle cytopenic myelofibrosis. Hematology ASH Education Program, 2022(1), 235–244. https://doi.org/10.1182/hematology.2022000340
Talpaz, M., & Kiladjian, J. J. (2021). Fedratinib, a newly approved treatment for patients with myeloproliferative neoplasm-associated myelofibrosis. Leukemia, 35(1), 1–17. https://doi.org/10.1038/s41375-020-0954-2
Tefferi, A., Cervantes, F., Mesa, R., Passamonti, F., Verstovsek, S., Vannucchi, A. M.,…Barosi, G. (2013). Revised response criteria for myelofibrosis: International Working Group-Myeloproliferative Neoplasms Research and Treatment (IWG-MRT) and European LeukemiaNet (ELN) consensus report. Blood, 122(8), 1395–1398. https://doi.org/10.1182/blood-2013-03-488098
Venugopal, S., & Mascarenhas, J. (2022). The odyssey of pacritinib in myelofibrosis. Blood Advances, 6(16), 4905–4913. https://doi.org/10.1182/bloodadvances.2022007524
Verstovsek, S., Gotlib, J., Mesa, R. A., Vannucchi, A. M., Kiladjian, J. J., Cervantes, F.,…Gupta, V. (2017). Long-term survival in patients treated with ruxolitinib for myelofibrosis: COMFORT-I and -II pooled analyses. Journal of Hematology & Oncology, 10(1), 156. https://doi.org/10.1186/s13045-017-0527-7
Verstovsek, S., Mesa, R., Gupta, V., Lavie, D., Dubruille, V., Cambier, N.,…Harrison, C. (2023). Momelotinib long-term safety and survival in myelofibrosis: Integrated analysis of phase 3 randomized controlled trials. Blood Advances, 7(14), 3582–3591. https://doi.org/10.1182/bloodadvances.2022009311